CDC Relaxes Vaccination Requirements Due to Shortage of Coronavirus Doses | The State

Main entrance to the headquarters of the Centers for Disease Control and Prevention in Atlanta, Georgia.
Photo:
Erik S. Lesser / EFE
The Centers for Disease Control and Prevention (CDC) They have modified their recommendations for vaccination against the coronavirus, given the shortage of supplies and obstacles to its distribution, to allow patients to use one of the two approved vaccine in the first dose, and the other in the second dose.
So far there are only two coronavirus vaccines approved for emergency use in the United States, that of Pfizer-BioNTech and Moderna.
CDC will allow “under exceptional circumstances” patients to start using one of them, and finish immunization with the other, at an interval that should not be longer than six weeks.
The country’s main health authority established the new guideline despite the fact that such a change has not been studied in clinical trials. The change in recommendations will give more flexibility to the delayed vaccination campaign, at a time when incoming president Joe Biden launched an ambitious plan to supply 100 million vaccines in its first 100 days.
“The agency’s intent is not to suggest that people do something different, but to give physicians flexibility for exceptional circumstances,” CDC spokeswoman Kristen Nordlund told The New York Times..
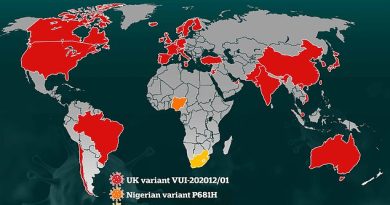
[La variante del coronavirus detectada en Reino Unido puede ser más mortal. Y preocupan otras]
This position is not unison. Dr. Anthony Fauci, special advisor to the Government in the fight against COVID-19, has repeatedly advised against making changes in the vaccination protocol without studies to support them.
The health authorities have also indicated that they will allow some flexibility within the prescribed period of 21 days, for the Pfizer vaccine, and 28, for the Moderna, between the first and second doses. “There is no maximum interval between the first and second doses for any of the vaccines,” says the CDC on its website.
[El intervalo de tiempo entre la primera y la segunda dosis de la vacuna no tiene que ser exacto, según expertos]
This room for maneuver has also been endorsed by national pharmacy chains, infectious disease experts, and doctors on the front line of medical care. Specialists argue that the body’s immune response does not expire after a set interval.
However, vaccine manufacturers’ guidelines indicate that two doses, given at specified intervals, are necessary for maximum protection against coronavirus.
Only for exceptions
The two vaccines licensed for emergency use, one from Pfizer and BioNTech and the other from Moderna, are based on the same method of achieving immunity, called mRNA, and require two doses.
Until now, CDC had followed the recommendations of its Advisory Committee on Immunization Practices, which specifically stated that vaccines should not be combined. The guide still states that the vaccines are “not interchangeable” and notes that the safety and efficacy of mixing doses has not been studied.
However, it now ensures that “in exceptional situations where the manufacturer of the vaccine for the first dose cannot be determined or where it is no longer available,” any available mRNA vaccine can be used in the second injection.
The new guideline also indicates that the time between one dose and another should be the closest to that recommended but, if this is not feasible, it can be extended to six weeks.
According to a senior official in the Biden Administration, pharmaceutical companies Pfizer and Moderna are on track to administer up to 18 million doses per week. Together, the companies have committed to delivering 200 million doses by the end of March.
With information from The New York Times.
.